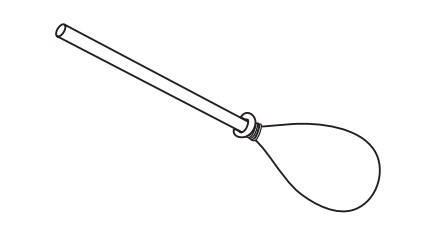
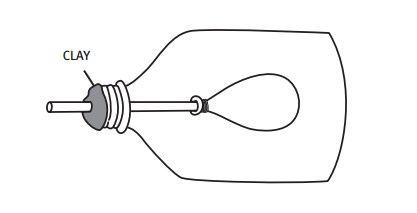
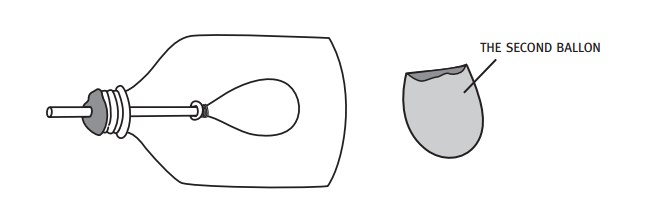
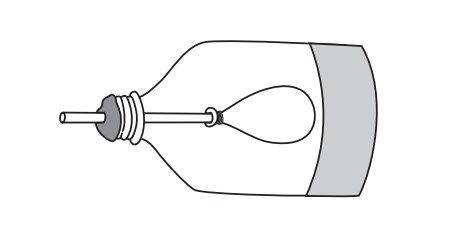
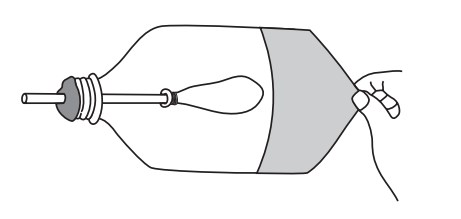
Students make a model of the human lung with a pop bottle, straw, and balloons.
"Winds will blow from high to low" applies even during breathing!
When we breathe in, the volume of the chest cavity is increased. Since there is more space available, the air pressure in the chest cavity and lungs is reduced, creating a low-pressure system in your body. The air outside your body immediately rushes in to equalize the lower pressure, flowing in through the nose or mouth into the lungs.
When we breathe out, the chest cavity contracts, increasing the air pressure in the lungs. The high-pressure air in the lungs rushes out of the nose and mouth to equalize the low-pressure zone of air around the body.
When the students pull the balloon diaphragm downwards they increase the volume of the model lung and therefore decrease the pressure inside. This causes the surrounding air to rush into the lung (going from high to low pressure), making the balloon lung inflate. The air in the balloon is trying to "fill in" the low-pressure zone inside the bottle. This mimics inhalation.
When the balloon diaphragm is pushed upwards, the volume of the bottle decreases, increasing the air pressure in the bottle. This causes the air in the bottle to push against the balloon, forcing the air out of it (high pressure air in the bottle wants to fill the low-pressure area outside). This mimics exhalation.